There are a range of definitions that describe nanoparticlesand the nanoscale. However, in general they describe particles that are 100nanometres (nm) or less in at least one dimension, and nanoscalemay be used to describe processes that take place at that scale. Theproperties of materials can be different at the nanoscale for two main reasons.Firstly, nanomaterials have a relatively larger surface area which can makematerials more chemically reactive. Secondly, particularly at lower sizeranges, quantum effects can begin to affect electron behaviour in the material.
Nanoremediation is the use of nanoparticles in remediation.Nanoremediation processes generally involve reduction or oxidation. In somecases this may be facilitated by an embedded catalyst. There is also someevidence that sometimes nanoremediation can work in combination with the insitu biological processes of dehalorespiration (where contaminants such assolvents are biologically dechlorinated).
The redox processes mediated by nanoremediation may lead tocontaminant degradation to simple compounds (for example in the treatment ofchlorinated solvents), or contaminant immobilisation in insoluble forms with limitedenvironmental accessibility (for example in the treatment of mobile arsenic)and/or reduced toxicity (for example in the conversion of chromium (VI) tochromium (III).
Nanoremediation is primarily deployed in situ in thesubsurface, in saturated zones. This is partly because of the contaminationproblems they have been developed to treat (FAQ: What are thepotential benefits of nanoremediation and its likely advantages over alternativetechnologies), and partly because the vast majority of nanoremediation hasused nZVI which tends to be rapidly passivated in aerobic environments. In mostcases nanoparticles are introduced via injection wells into an aquifer,typically requiring some form of active pumping.
There are two modes of deployment of in situremediation: control of the source term and control of the pathway. These maybe applied individually or together to break source-pathway-receptor linkages.These modes of deployment relate to the general principles of how risks fromcontaminated land are assessed and managed for remediation in general see RBLM on COMMON FORUMweb site.
- Pathway management describes the use of nanoremediation to degrade or immobilise contaminants migrating along pathways, typically being carried by groundwater flow from the source term.
- Source reduction describes the use of nanoremediation to treat a source term, for example a NAPL source within the saturated zone. The intention is usually to reduce the mass of the source term so that the duration of groundwater contamination is decreased. Note: In common with all in situ remediation techniques, in situ source term treatments may render residual sources more mobile and so lead to higher contamination fluxes to groundwater. Success is crucially dependent on the accessibility of the source term to the treatment being deployed, and this may limit applicability.
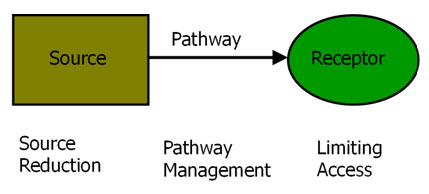
Remediation interventions for risk management

Direct Injection of nZVI in the Field at the Trenton Facility, New Jersey (Photo courtesy of Geosyntec Consultants)
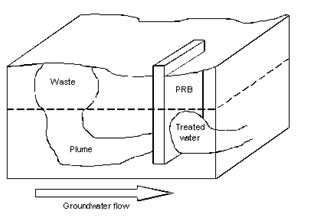
Typical PRB Configuration (from www.readyreference.co.uk)
Nano-scale zero valent iron particles (nZVI) have been themost extensively used approach in nanoremediation projects to date. Asproduced, most nZVI tested falls into the 10-100 nm size range. See FAQ: "Where havenanoparticles been used in remediation?" See NanoRem ToolBox,Bulletins (http://www.nanorem.eu/toolbox/bulletin-shelf.aspx#TB1).
nZVI is a highly reactive reducing agent and as suchdegrades some organic compounds (e.g. chlorinated solvents) and changes theoxidation state of elements (e.g. chromium, uranium). This underlies itsfunctionality in soil and groundwater remediation. nZVI is unstable in air andin the ground environment and is rapidly inactivated by several mechanisms. Anumber of modifications have been developed to improve stability and/orfacilitate the migration of nZVI in aquifers (by coating and/oremulsification). nZVI particles have also been developed which are doped withlow levels of other metals (in particular palladium) which are catalytic andincrease their reactivity. However, typically there will always be a trade-offbetween reactivity and stability and between reactivity and mobility in thesubsurface.
As well as nZVI there are emerging classes of nanoparticlesthese include:
- Oxidising agents, such as those based on oxides of iron
- Nanoparticles which combine sorption and redox, for example, to provide a more effective treatment solution of nonaqueous phase liquids
- Nanoparticles that are anchored or embedded in a matrix, for example, for use in permeable reactive barriers
A wide range of nanoparticles are being investigated by theNanoRem project (See NanoRem Tool Box, Nanoparticles and Tools (http://www.nanorem.eu/toolbox/nanoparticles_and_tools.aspx#TB1).
Further Information
Related Thematic Pages
Related FAQs: